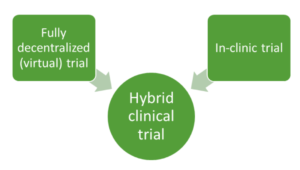
Hybrid clinical trials to generate real-world evidence: design considerations from a sponsor's perspective - ScienceDirect
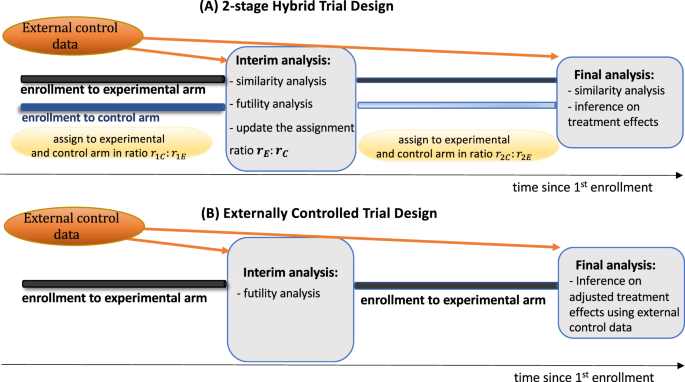
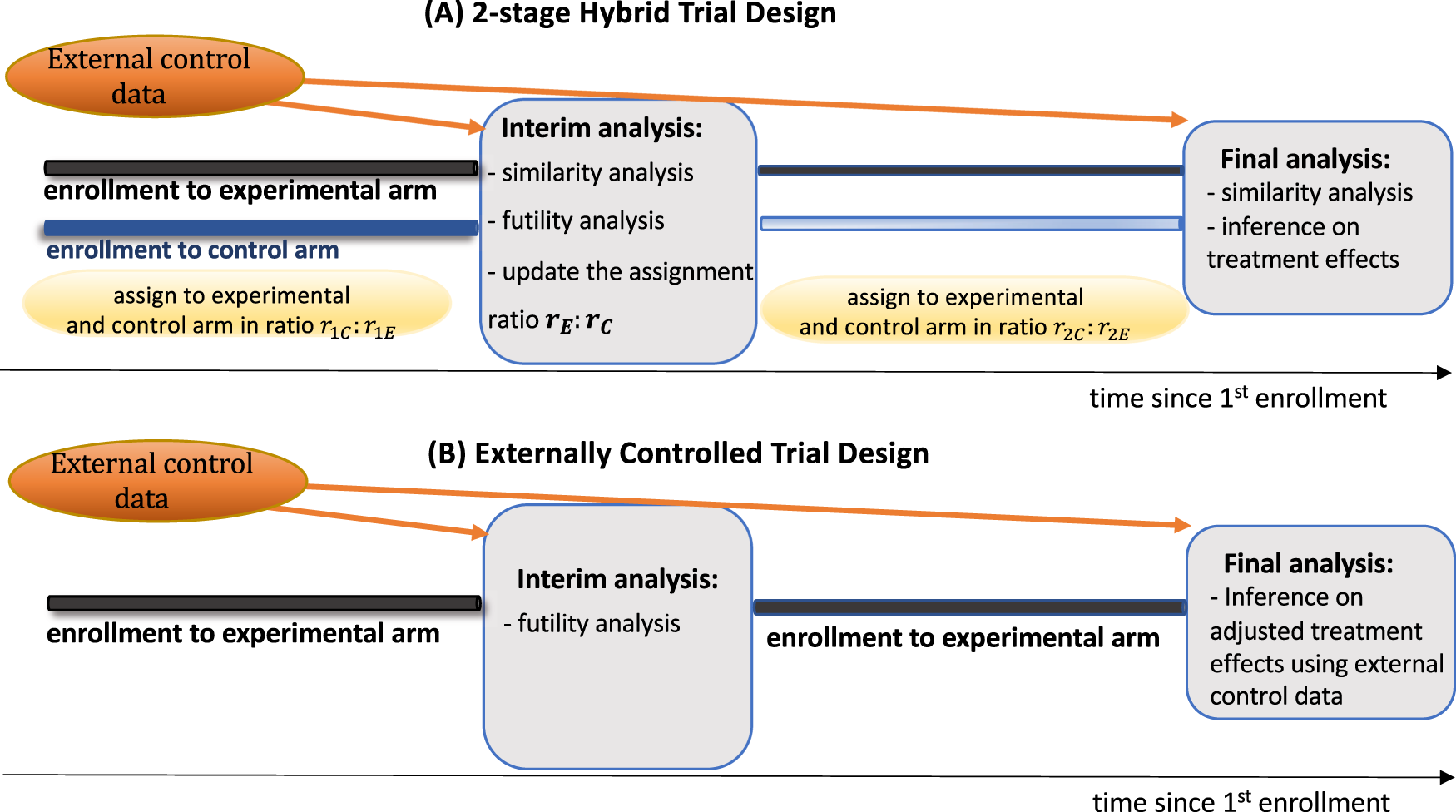
The design and evaluation of hybrid controlled trials that leverage external data and randomization | Nature Communications
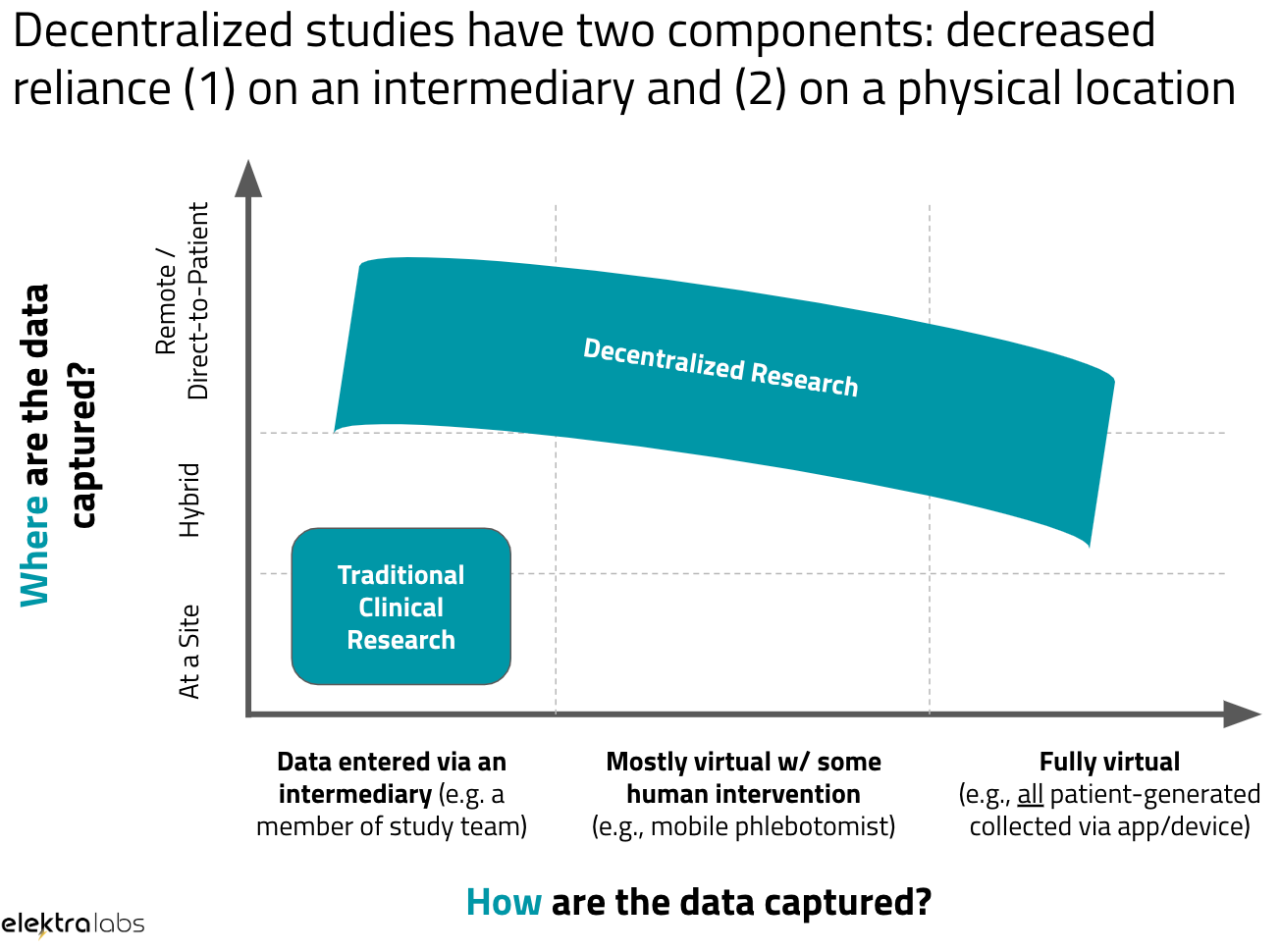
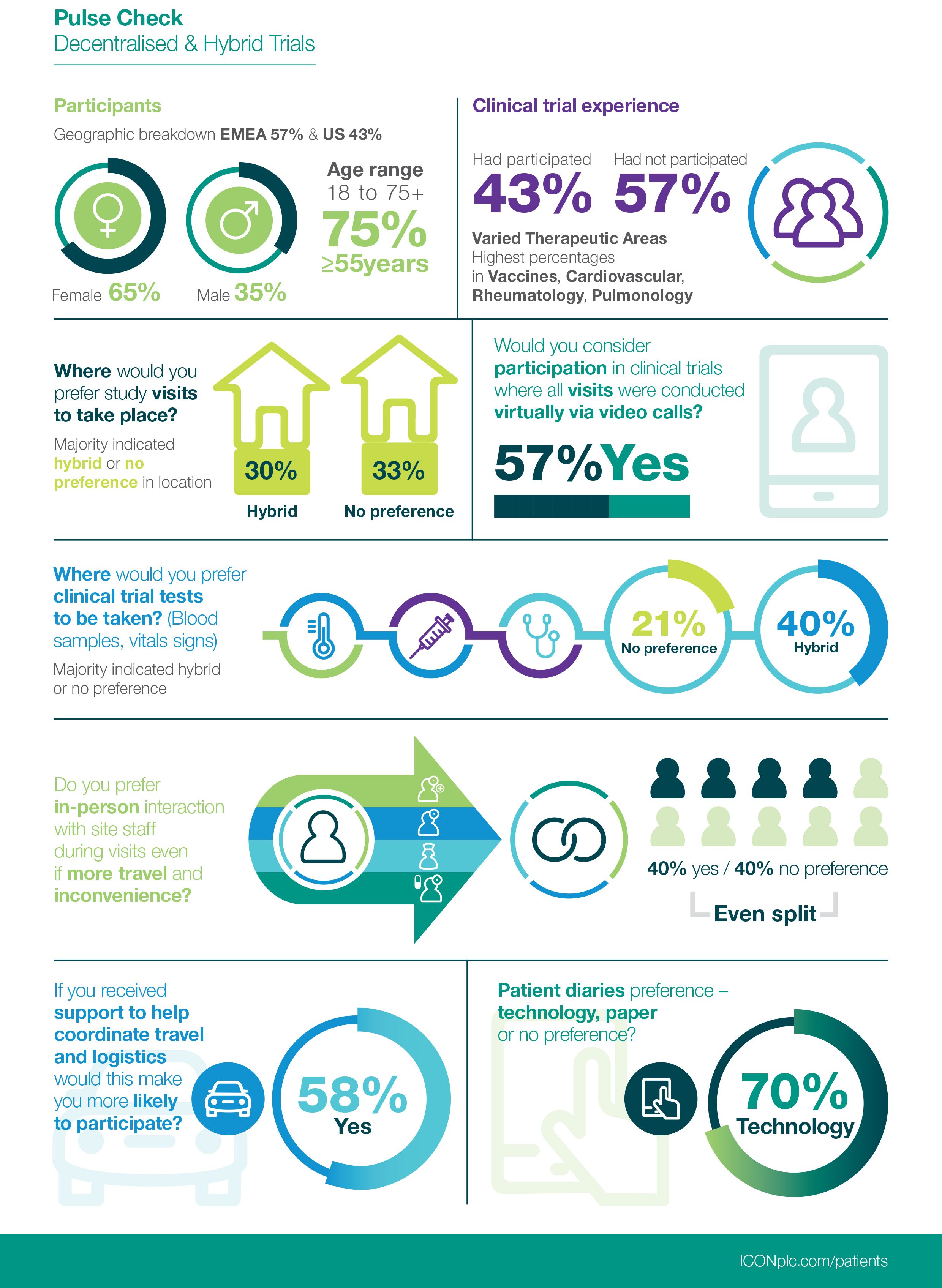
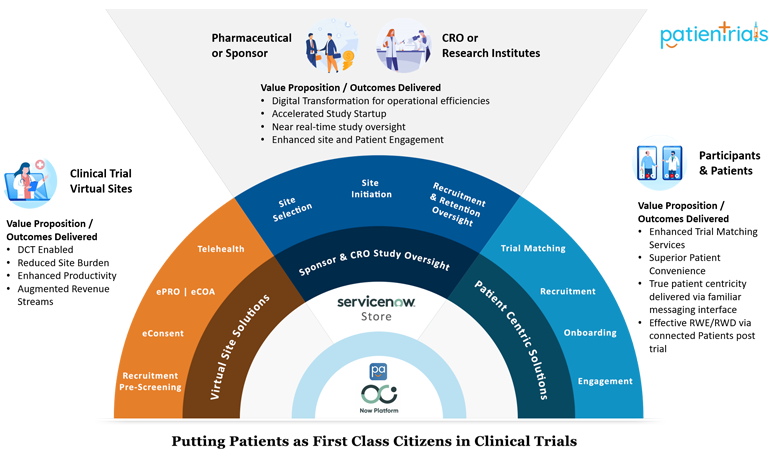
Decentralized clinical trials. A framework for “direct-to-patient,”… | by Andy Coravos | HumanFirst | Medium