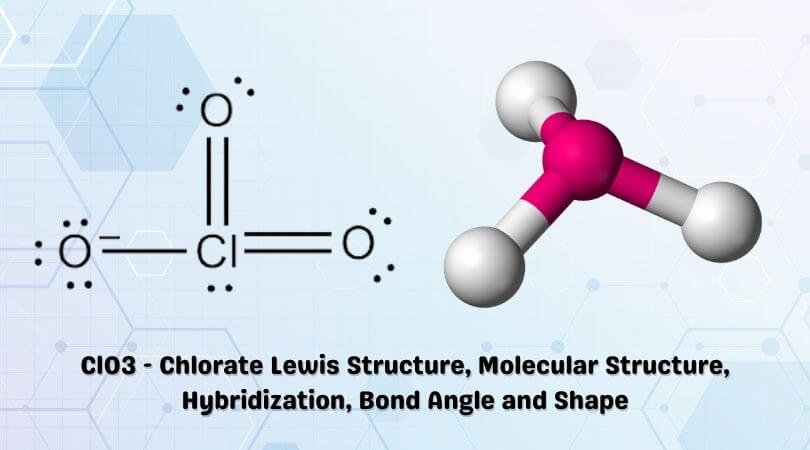
Hybridization of Chlorine in Hypochlorite ClO-, Chlorite ClO2-, Chlorate ClO3-, Perchlorate ClO4- - YouTube
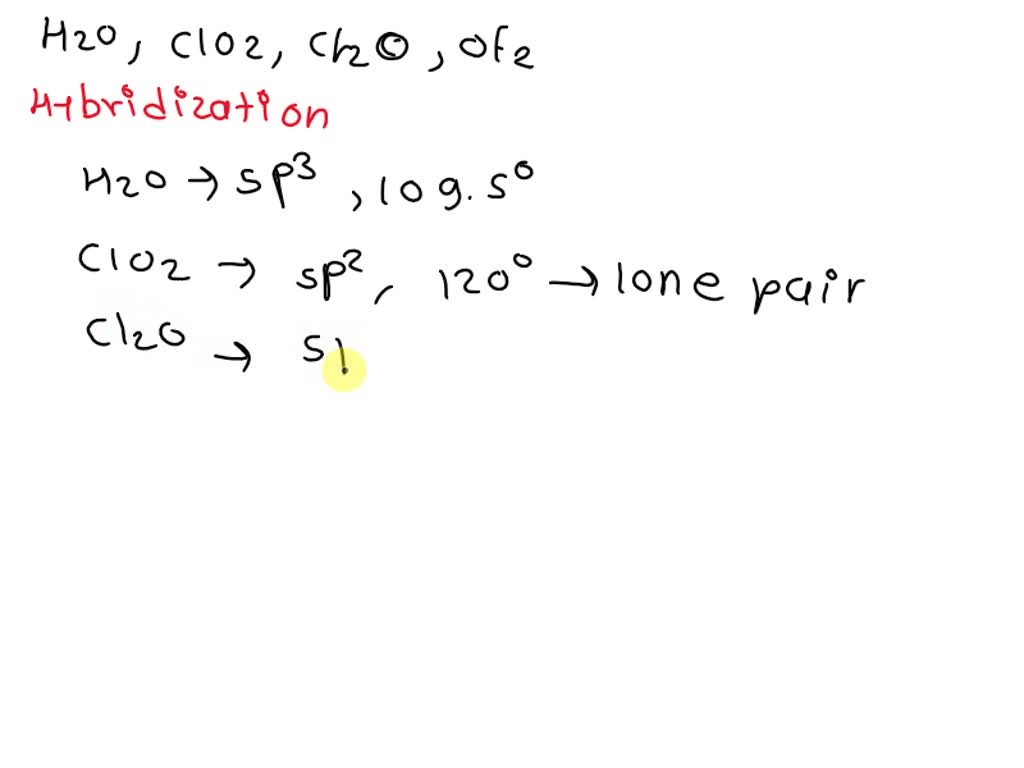
SOLVED: Determine the increasing order of bond angles in the following compounds: (i) H2O (ii) ClO2 (iii) ClO (iv) OF2. (A) H2O < ClO2 < ClO < OF2, (B) ClO2 < ClO <
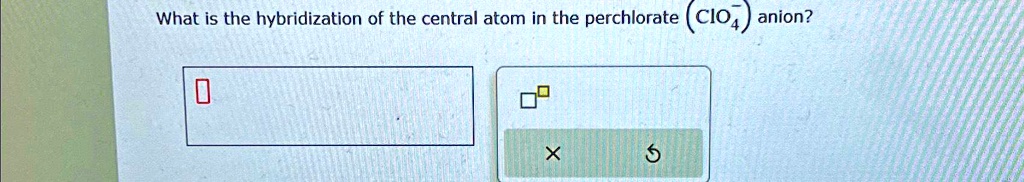
SOLVED: What is the hybridization of the central atom in the perchlorate ( ClO(4)^(-))anion? ◻◻ What is the hybridization of the central atom in the perchlorate (CIO4) anion? 5

Qtion no 7​​ 7 Correct statement(s) for ClO4- is (A) Total number of electron taking part - Chemistry - Solutions - 12713323 | Meritnation.com

Hybridization of Chlorine in Hypochlorite ClO-, Chlorite ClO2-, Chlorate ClO3-, Perchlorate ClO4- - YouTube

Hybridization of Chlorine in Hypochlorite ClO-, Chlorite ClO2-, Chlorate ClO3-, Perchlorate ClO4- - YouTube

The hybridization of Cl atom in ClO_(4)^(-) and ClO_(3)^(-) is | CLASS 12 | NONE | CHEMISTRY | D... - YouTube

What is the steric number of ClO- ? Specify and draw ClO- electron pair geometry. What is ClO- hybridiztion | Homework.Study.com